Liters = 5 kg ÷ 0.7 kg/L = 7.1429 L When to Convert Kilograms to Liters Thus, the volume in liters is equal to the weight in kilograms divided by the density (in kg/L) of the ingredient, substance, or material. You can use this simple formula to convert: Note that in order for this to work, the density must be in kilograms per liter (kg/L). To convert a measurement in kilograms to liters, divide the weight by the density of the ingredient or material. Therefore, to convert between kilograms and liters of an ingredient or substance, we must either multiply or divide by its density, depending on which direction we are performing the conversion. 
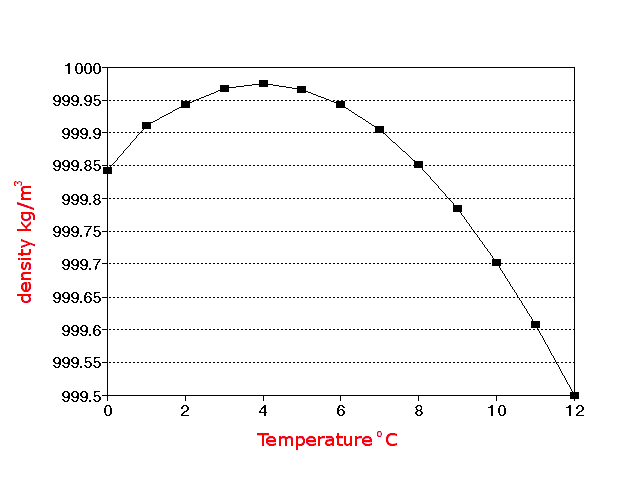
In this case, we need to account for the density of the substance whenever we do a conversion. Since kilograms are a unit of mass and liters are a unit of volume, which are different physical quantities, we need to know one more physical quantity of the ingredient or substance to convert between them. He specializes in math, science, and astrophysics. Thermal properties of water at different temperatures like density, freezing temperature, boiling temperature, latent heat of melting, latent heat of evaporation, critical temperature and more.Ethan has a PhD in astrophysics and is currently a satellite imaging scientist. Online calculator, figures and tables showing Specific Volume of water at temperatures ranging from 0-370 ☌ and 32 - 700 ☏ - Imperial and IS Units. Temperatureįigures and tables showing the enthalpy and entropy of liquid water as function of temperature - SI and Imperial Units. Water - Density, Specific Weight and Thermal Expansion Coefficientsĭefinitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360☌ (32 to 680☏). 
Properties of the US standard atmosphere ranging -5000 to 250000 ft altitude. The definition of STP - Standard Temperature and Pressure and NTP - Normal Temperature and Pressure. STP - Standard Temperature and Pressure and NTP - Normal Temperature and Pressure Specific volume of moist air is defined as the total volume of humid air per mass unit of dry air Water Content and Temperatureĭensity of the mix of dry air and water vapor - moist humid air. Pressure, temperature and volume in a perfect ideal gas like moist air (air with water vapor).ĭensity of moist air vs. Online density converter with commonly used units. Thermal properties of air at different temperatures - density, viscosity, critical temperature and pressure, triple point, enthalpi and entropi, thermal conductivity and diffusivity and more. Online calculator, figures and tables showing density, specific weight and thermal expansion coefficients of air at temperatures ranging -100 to 1600 ☌ (-140 to 2900 ☏) at atmospheric and higher pressure - Imperial and SI Units.ĭry air is a mixture of gases where the average molecular weight (or molar mass) can be calculated by adding the weight of each component. 
Pressure and TemperaturesĪir density at pressure ranging 1 to 10 000 bara (14.5 - 145000 psi) and constant selected temperatures.Īir - Density, Specific Weight and Thermal Expansion Coefficient vs. Altitudeĭensity and specific volume of air varies with elevation above sea level.Īir - Density vs. Definitions and convertion calculators.Īir - Density and Specific Volume vs. Moist and humid air - psychrometric charts, Mollier diagrams, air-condition temperatures and absolute and relative humidity and moisture content.ĭensities of solids, liquids and gases. Dry Air and Water Vapor - Density and Specific Volume vs. Values are for atmospheric pressure - 29.96 inches Mercury (760 mm Mercury, 14.696 psia, 1.0333 kg/cm 2, 101.33 kPa ). Density and specific volume of dry air and water vapor at temperatures ranging 225 to 900 o F (107 to 482 o C) are indicated below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
